Lung transplantation can be lifesaving for some people with severe chronic lung conditions. Unfortunately, the body’s innate immune response often causes damage to donor lungs. A new study has identified triggers that cause tissue damage in donor lungs during reperfusion. Targeting these triggers with future therapies could improve lung transplant outcomes and long-term function by reducing the risk of ischemia-reperfusion injury.
The study, led by investigators in the Thoracic Immunology Laboratory at Washington University School of Medicine in St. Louis, was published March 1 in the journal Proceedings of the National Academy of Sciences.
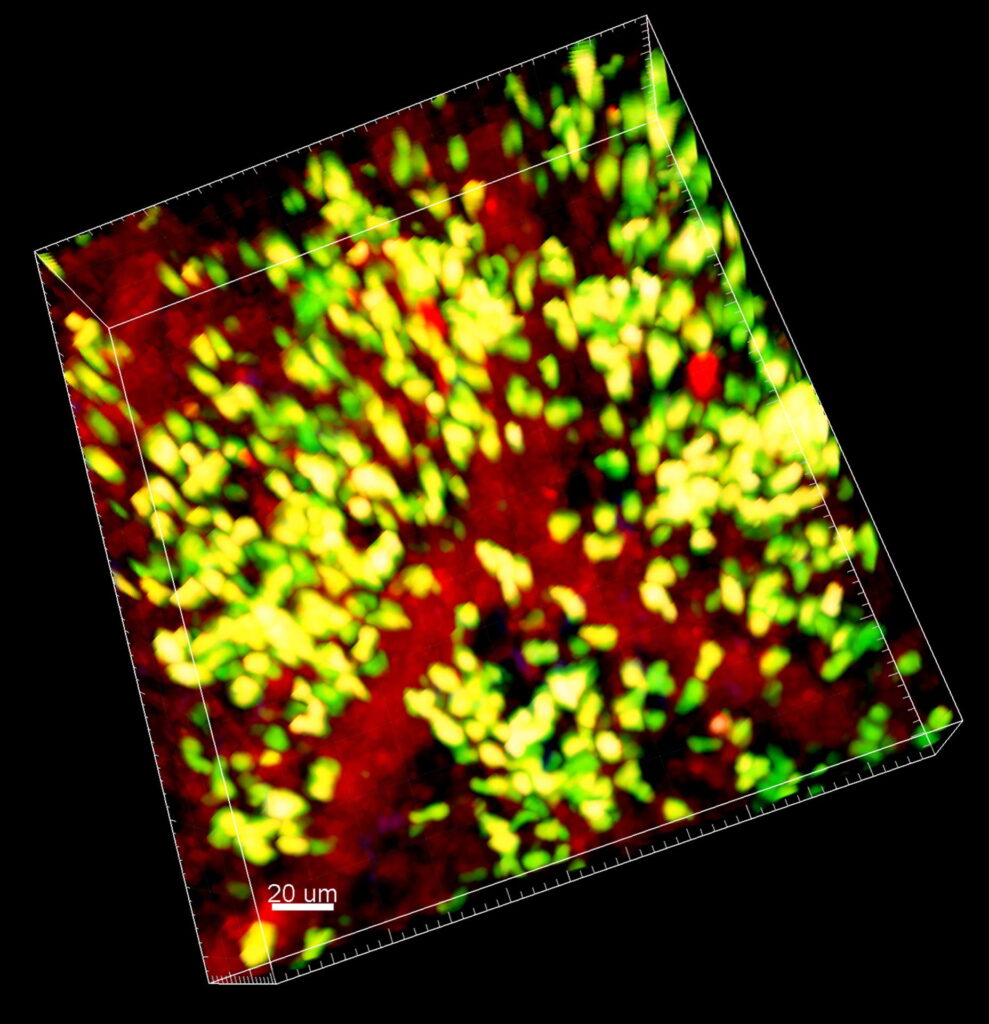
“We found that there is substantially more infiltration of neutrophils in the lungs when lungs are subjected to a period of warm ischemia. These neutrophils are attracted to certain vessels, which they destroy on their way into the air spaces. This process is associated with substantial damage to the donor lungs. We were able to describe the individual steps that guide the neutrophils into the lungs during this process,” says first author Wenjun Li, MD, an associate professor of surgery and a member of the Thoracic Immunology Laboratory.
Neutrophils are a type of white blood cell that help the body fight infection. Normally, there are a small number of neutrophils in the lungs to defend against pathogens.
Before an organ is transplanted, it typically goes through a period without blood flow or oxygen, called ischemia. During organ transplantation blood flow is restored through the reperfusion process. Restoring blood flow—which is vital for the success of the organ—can lead to ischemia-reperfusion injury.
The investigators described that necroptosis, a type of inflammatory regulated cell death in the lungs, triggered recruitment of neutrophils. “The damage caused by necroptosis releases damage-associated molecular patterns, which engage receptors on the cells in the lungs, including nonclassical monocytes and vascular endothelial cells. These nonclassical monocytes secrete chemokines, which bring neutrophils into the vessels. Think of chemokines as the immune system’s breadcrumbs. Neutrophils follow these breadcrumbs to the vessels, where they cause sterile inflammation. The neutrophils destroy the vessel wall and lead to lung graft dysfunction,” says study senior author Daniel Kreisel, MD, PhD, the inaugural G. Alexander Patterson, MD/Mid-America Transplant Endowed Distinguished Chair in Lung Transplantation.
Kreisel, a professor of surgery, pathology and immunology, and Li, an associate professor of surgery, used intravital imaging to observe how the immune system responds to lung transplantation in real time. Intravital imaging allows researchers to see biological processes in vivo at a high resolution. This technique makes it possible to see how living cells behave in their natural environment. These contributions by investigators from The Thoracic Immunology Laboratory, some recent and some published over decades of study, have advanced the scientific understanding of immune responses to transplantation for researchers around the globe.
One key finding from this study is that the lungs are damaged through a different process than other organs. There are different forms of inflammatory cell death. In 2019, Li published a study describing the inflammatory cell death pathway affecting donor hearts. Ferroptosis, another type of cell death, triggers inflammation after heart transplantation, which differs from the necroptosis in transplanted lungs described in the PNAS study.
The ultimate goal of this field of research is to prevent or reduce inflammatory cell death in donor organs. If the cells do not die, the cascade that causes ischemia-reperfusion injury and primary graft dysfunction is not initiated. Li notes that, in the study, blocking necroptosis resulted in very little inflammation.
“With this understanding of the form of cell death affecting lungs during ischemia, we can target necroptosis with future therapeutics,” says Kreisel, who is surgical director of the Lung Transplant Program at Barnes-Jewish Hospital. “Tailoring future pharmacological therapies for each organ could prevent inflammatory responses in transplanted organs, meaning a reduced risk of ischemia-reperfusion injury and primary graft dysfunction.
“One take-home message of this study is that each organ has its own intricacies in terms of immune response and cell death pathways,” Kreisel says. “Therapies need to be tailored so that we treat lung transplantation with different drugs than other organs.”